*The search scope is limited to the products and news section of this site

Recently, the 2026 American Association for Cancer Research Annual Meeting (AACR 2026) was held in San Diego, USA. At this meeting, Genuine Biotech presented preclinical findings of Azvudine (FNC) combined with Doxitinib in the form of a poster. In preclinical studies of EGFR-mutant non-small cell lung cancer (NSCLC), significant synergistic anti-tumor effects were observed, providing a promising new regimen to overcome EGFR-tyrosine kinase inhibitor (EGFR-TKI) resistance.
Title: Azvudine Combined with Doxitinib, a Potential Therapy for EGFRm+ NSCLC
Time: 2:00 PM – 5:00 PM, Apr. 21, 2026 (Pacific Time)
Location: Poster Section 42
Currently, EGFR-TKIs represented by osimertinib are the standard therapy for patients with EGFR-mutant NSCLC. However, both intrinsic resistance and acquired resistance remain key bottlenecks limiting treatment efficacy. Based on this unmet clinical need, Genuine Biotech is advancing the combination regimen of FNC and Doxitinib.
1. Combined antitumor mechanisms
As a highly selective nucleoside drug with a unique dual mechanism, FNC shows marked potential in arresting tumor growth. It not only inhibits tumor cell proliferation by terminating DNA chain elongation and disrupting enzymes involved in tumor cell nucleic acid synthesis, but also modulates the tumor immune microenvironment (TIME) by suppressing myeloid-derived suppressor cell (MDSC) differentiation and infiltration while enhancing CD8⁺ T cell and NK cell activation, thus shifting the TIME from immunosuppressive to immune-activated, thereby overcoming EGFR-TKI resistance at the mechanistic level.
Doxitinib is a highly potent and selective, orally administered epidermal growth factor receptor (EGFR)-targeted drug candidate independently developed by Genuine Biotech. Preclinical studies have shown that Doxitinib can reduce toxic metabolite levels by up to 80% compared with osimertinib. It also demonstrated a favorable safety profile in patients with EGFR mutations in the Phase 1/2a clinical study, and encouraging clinical efficacy was observed in patients with brain metastases, laying the foundation for subsequent combination therapy.
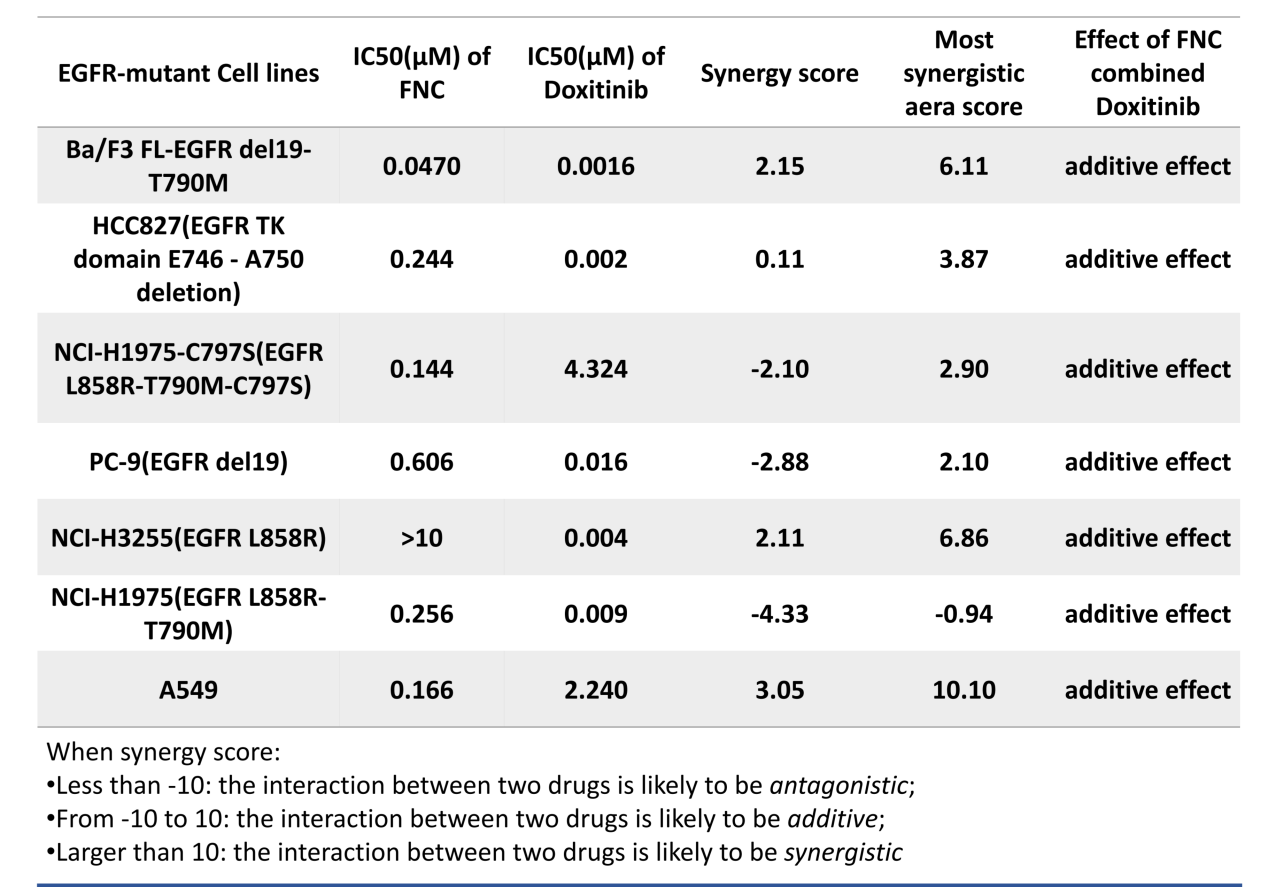
2. In vitro studies have shown synergistic anti-proliferative effects
In vitro studies showed that FNC and Doxitinib exhibited additive anti-proliferative effects in various EGFR-mutant NSCLC cell lines, including L858R/T790M/C797S triple-mutant cells, providing direct cellular-level evidence for combination therapy.
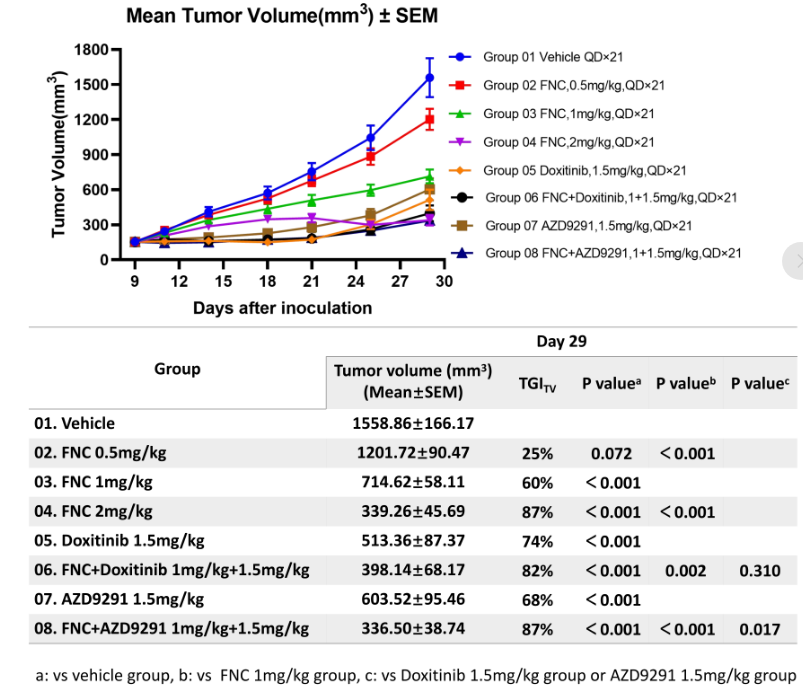
3. Superior tumor growth inhibition observed in the NCI-H1975 (EGFR L858R/T790M) subcutaneous xenograft model
The combination of FNC and Doxitinib showed superior tumor growth inhibition compared to either monotherapy in the NCI-H1975 (EGFR L858R/T790M) subcutaneous xenograft model. Among them, the tumor growth inhibition (TGI) of the FNC 1 mg/kg + Doxitinib 1.5 mg/kg group reached 82%, which was significantly superior to that in the Doxitinib monotherapy group, and the safety profile was favorable, providing strong support for subsequent clinical translation.
In Sep. 2025, FNC combined with Doxitinib was approved by the National Medical Products Administration for the conduct of clinical trials in patients with EGFR-mutant locally advanced or metastatic NSCLC. In Nov. 2025, the first participant was enrolled and dosed, and the Phase 1/2a clinical study was officially initiated.
In the future, Genuine Biotech will continue to promote the clinical translation of this combination therapy, while addressing unmet clinical needs, and relying on its independent R&D platform to explore more innovative tumor treatment options to bring new treatment options to patients worldwide.
